Olympus Launches THUNDERBEAT Energy Device for Open Surgery
September 28, 2022
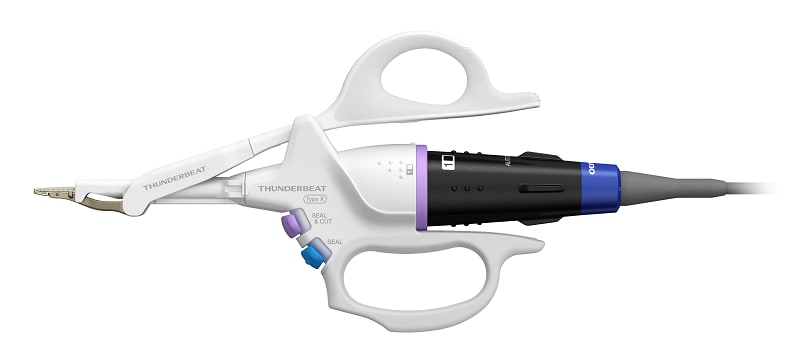
THUNDERBEAT™ Open Fine Jaw Type X
TOKYO and CENTER VALLEY, PA., (September 28, 2022) - Olympus Corporation (Olympus), a global medtech company committed to making people’s lives healthier, safer, and more fulfilling, today announced the release of the THUNDERBEAT™ Open Fine Jaw Type X surgical energy devices for open surgery. With a new thermal shield, the THUNDERBEAT Open Fine Jaw Type X surgical energy device is designed to support safer procedures.1 The device is available commercially in Japan. The commercial launch of the product in Europe, the U.S., and South Korea is expected in October 2022, with continued launches in other countries and regions following.
This device is part of Olympus’ THUNDERBEAT portfolio of hybrid energy devices that deliver both ultrasonic and bipolar energy simultaneously for tissue management, including hemostatic cutting and dissection, in laparoscopic surgery and open surgery. The THUNDERBEAT hybrid devices eliminate the need for multiple instruments during the surgery, contributing to efficiency in the operating room and reduced operation time.2
“This latest addition to our differentiated THUNDERBEAT line of products continues to deliver on Olympus’ commitment to offering hospitals a full portfolio of Advanced Energy devices providing best-in-class functionality for numerous procedures and specialties in minimally invasive and open surgery,” said Phil Roy, Global Vice President and General Manager of the Surgical Devices Business Unit.
The THUNDERBEAT™ Open Fine Jaw Type X device is designed for open surgical procedures that require delicate and fine tissue dissection, such as in thyroidectomy or radical neck dissection. The new thermal shield improves the thermal profile on the grasping surface, which reduces the risk of unintended heat damage to nearby tissue, nerves and other structures.1 The jaw design maintains a finely curved tip for precise dissection and enhanced visibility during use.1
New Thermal Shield Supports Safer Procedures
The newly developed thermal shield at the distal tip slows heat transfer from the probe to the exterior surface of the jaw and reduces the risk of unintended heat damage to surrounding tissue and vessels.1
Fine Shape of Distal Tip Supports Precise Procedures
The slim shape of the distal tip supports precise tissue management capabilities including fine dissection and firm grasping force, cutting and sealing to the tip of the device, and blunt dissection.1
Simultaneous Output of Two Energies Enables a Variety of Operations with One Device
Delivering simultaneous bipolar and ultrasonic energy enables fast, hemostatic cutting, easy dissection, and reliable vessel sealing and division. A separate advanced bipolar1 function enables vessel sealing and spot coagulation without cutting, when desired.
1 Data on file with Olympus as of May 11, 2020
2 Data on file with Olympus as of June 26, 2015
The THUNDERBEAT™ Open Fine Jaw Type X device is a single use hybrid ultrasonic and bipolar electrosurgical instrument intended for use in open surgery. This device should not be used for tubal sterilization or tubal coagulation for sterilization procedures. Use with caution in patients with electronic implants, such as a cardiac pacemaker, or nerve simulators, to avoid possible hazard to patients due to interference. Before use, thoroughly review the product manual and use the equipment as instructed.
The THUNDERBEAT Open Fine Jaw Type X device is manufactured by Olympus Medical Systems Corporation.
About Olympus
Olympus is passionate about creating customer-driven solutions for the medical, life sciences, and industrial equipment industries. For more than 100 years, Olympus has focused on making people’s lives healthier, safer and more fulfilling by helping to detect, prevent, and treat disease; furthering scientific research; and ensuring public safety.
In its Therapeutic Solutions business, Olympus uses innovative capabilities in medical technology, therapeutic intervention, and precision manufacturing to help healthcare professionals deliver diagnostic, therapeutic, and minimally invasive procedures to improve clinical outcomes, reduce overall costs, and enhance the quality of life for patients. Starting with its early contributions to the development of the polypectomy snare, Olympus’ Therapeutic Solutions portfolio has grown to include a wide range of medical devices to help prevent, detect, and treat disease. For more information, visit www.olympus-global.com and follow our global Twitter account: @Olympus_Corp.
* The contents in this website including products availability, specifications or prices are the information as of the date of announcement and are subject to change without prior notice.
* Information is intended to be presented to the media, shareholders, investors, and other interested parties. Information about our medical products (including products currently under development) included in this website is not intended for advertising or medical advice.
* Olympus Corporation assumes no responsibility for any damage resulting from the use of this material.
* All company names and product names mentioned in this website are trademarks or registered trademarks of their respective companies. ® and TM marks are not specified in this website. All trademarks and registered trademarks are the property of their respective owners.

